 The positive oxidation states allow transition elements to form many different ionic compounds. 
Some just lose one electron to form +3 cations, while others form even higher charges. Many of the transition metals can lose two or three electrons, forming cations with charges of +1 or +2. The transition elements do have low ionization energies, however, so they exhibit a wide range of oxidation states. Unlike their alkali and alkaline earth neighbors, most transition metals can be found in nature in their neutral state. They'd stay in bed all day if we let them. And electrons are lazy-they'd rather not exert extra energy if they don't need to. Just know that filled or half-filled orbitals save energy and everyone loves to save energy. At this point in your chemistry career it's easier to accept the fact there are exceptions to every rule. We'd love to give you a quick and easy-to-understand explanation for these exceptions, but the reasoning is complicated, confusing, and uber-difficult. The key is to look for opportunities to achieve a filled or half-filled d-shell. There are quite a few exceptions when filling the aufbau diagram across the transition metal series. This means the actual valence electron configuration of palladium is: Remember, the d-orbital can hold a total of 10 electrons. This seems reasonable, but if we do a quick electron shuffle and put the two s-electrons into the d-orbital, we achieve a super stable electron configuration because the d-shell is completely filled. What's the valence electron configuration? If we just follow the diagram we get: (full electron configuration) Ti: 1s 2 2s 2 2p 6 3s 2 3p 6 4s 2 3d 2 (valence electron configuration) Ti: 4s 23d 2 Let's check out some examples.įirst titanium, easy peasy lemon squeezey. Electrons like to have the lowest possible energy-that's how they roll. The general rule is to follow the above diagram, but if there is an instance where a quick shuffle of electrons can result in a completely filled or half-filled orbital, the electrons will tend to do the electron shuffle. The transition metals are located in the d-block so their valence electrons must go into d-orbitals, right? Yes and no. The alkali and alkaline earth groups are in the s-block so their valence electrons went into s-orbitals. But where do these electrons go? Let's check out an aufbau principal diagram. Care to wager how many valence electrons group 12 has? We'll give you a hint: It starts with 1 and ends with 2. Group 11 elements have 11 valence electrons. Group 10, for example, has 10 valence electrons. It's an instant assistant on valence electron counts. The number of the group in which a metal resides is important and can be a huge aid on quizzes and exams. 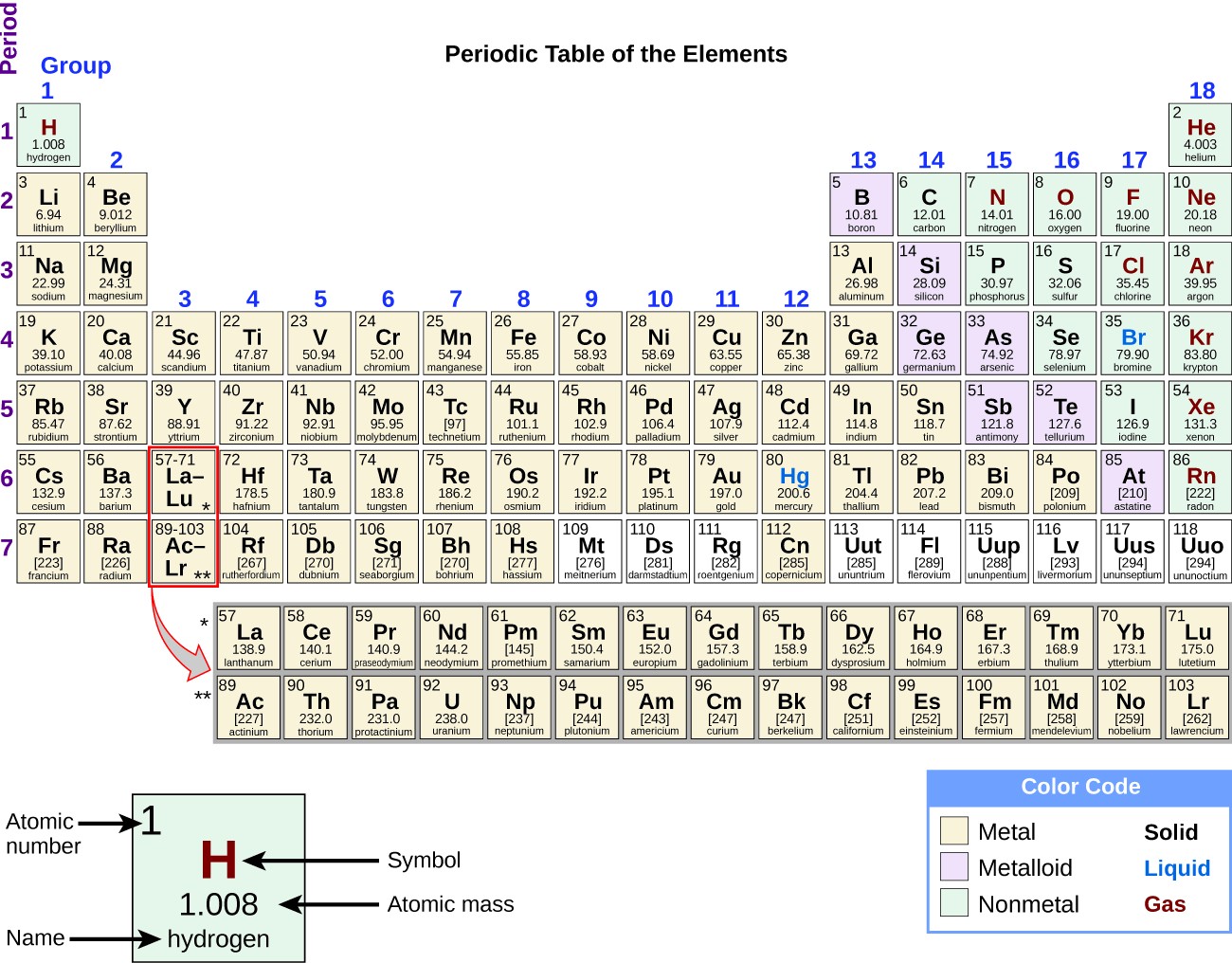
Think this information is useless? You might end up on Jeopardy! some day and then you'll be thanking us. Rounding up the transition metals are group 12 which include zinc (Zn), cadmium (Cd), and mercury (Hg). 
A jeweler's favorite group 10 is home to the elements of copper (Cu), silver (Ag), and gold (Au). The group 10 members are nickel (Ni), palladium (Pd), and platinum (Pt). Cobalt (Co), rhodium (Rh), and iridium (Ir) make up group 9. Group 8 is comprised of iron (Fe), ruthenium (Ru), and osmium (Os). Group 7 is home to manganese (Mn), technetium (Tc), and rhenium (Re). We certainly can't forget about the group 6 members of chromium (Cr), molybdenum (Mo), and the ever-tricky tungsten (W). Group 5 is comprised of vanadium (V), niobium (Nb), and tantalum (Ta). The group 4 elements are titanium (Ti), zirconium (Zi), and Hafnium (Hf). The group 3 elements are scandium (Sc) and Yttrium (Y). These d-block elements are the short columns in the center of the periodic table from group 3 to group 12. Transition Metal MadnessLocated in the heart of the periodic table are the ever-favorite transition metals, also known as the transition elements. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
